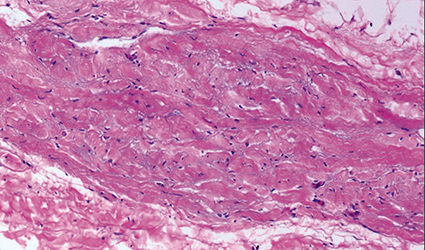
A 12-year-old boy from Colorado had a rare disease which hardened his skin like a stone.
His father first took him to see a doctor after he noted a hard lump on the boy's right thigh. Later he was diagnosed with stiff skin syndrome. The painful disease kept spreading, to his stomach, hips and his back. Later it spreads to his chest area, caused breathing issues.
Stiff skin syndrome causes a person's skin to harden and thicken across the entire body. This thickening results in a person being unable to move his or her joints, leaving the joints stuck in a bent position. The genetic disorder affects the protein named fibrillin-1, which allows the elastic fibers in the skin to stretch.
This progressive disease usually affects the shoulders, knees and elbows and could also make it difficult to bend the fingers. Other symptoms include loss of body fat, excessive hair growth, scoliosis, slow growth, muscle weakness and short stature.
At present, there are no treatments that could reverse the symptoms of stiff skin syndrome. Only physical therapy could bring some relief in joint-movement issues.
Moreover, there is evidence that an immunosuppressive drug called mycophenolate mofetil may improve symptoms if administered along with physical therapy.
The post Boy’s Skin becomes hard as stone –Stiff Skin Syndrome appeared first on Drugdu.com
from Drugdu https://goo.gl/QgQoHk